Opioid-addicted baby at center of class-action lawsuit in Southern Illinois
The family of a baby born addicted to opioids filed a class-action lawsuit Wednesday against 20 pharmaceutical companies they accused of being negligent and responsible for the mother's addiction to opioids and heroin.
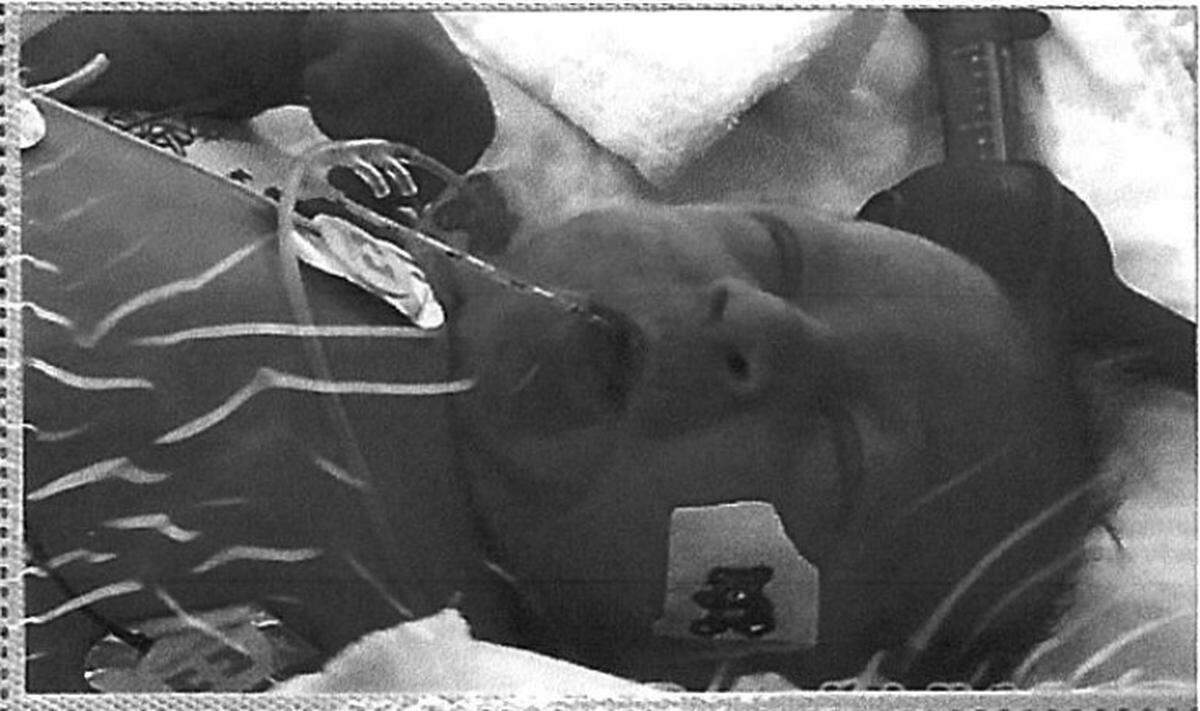
The lawsuit tells the story of a baby, only identified by his initials T.W.B. He was born March 21, 2017, and spent the first days of his life in "excruciating pain" as he was weaned from his opioid addiction, inherited from his mother.
"Baby T.W.B. will require years of treatment and counseling to deal with the effects of prenatal exposure," the lawsuit says. "Baby T.W.B. and his mother are victims of the opioid crisis that has ravaged Illinois, causing immense suffering to those born addicted to opioids and great expense to those forced to deal with the aftermath."
Alton residents Deric and Ceonda Rees, the baby's grandparents, filed the lawsuit Wednesday in the U.S. District Court of Southern Illinois. Ceonda Rees declined to comment, saying she had been advised not to discuss the lawsuit.
The baby was diagnosed with Neonatal Abstinence Syndrome, which can cause breathing and feeding problems, and require a longer hospital stay while they are experiencing withdrawal symptoms.
Taking opioids during pregnancy can also cause babies to be born with birth defects, according to Stanford Children's Health. The number of babies born with this disease has increased fivefold since 2000, according to the National Institute on Drug Abuse.
At birth, Baby T.W.B. shook, refused to feed and cried excessively as he arched his back from the pain, according to the lawsuit. It was difficult to soothe him, and his development was delayed.
The baby's mother became addicted to opioids when she was prescribed them for a broken ankle and broken hand as a young teenager. Her addiction to prescription opiates functioned as a gateway to heroin addiction, according to the lawsuit. The lawsuit alleges the mother consumed drugs made and distributed by each of the 20 defendants, including Dilaudid, Percocet, Oxycodone and Hydrocodone.
The lawsuit accuses the pharmaceutical companies of creating an environment where opioids freely flow due to "false, negligent and unfair marketing/unlawful diversion of prescription opioids."
The Reeses asked for equitable relief and medical monitoring to identify developmental issues that will "almost inevitably appear as they age," and fund services and treatment.

"Baby T.W.B.'s experience is part of an opioid epidemic sweeping through the United States, including Illinois, that has caused thousands of infants great suffering and continuing developmental issues," the lawsuit says." This epidemic is the largest health care crisis in U.S. history."
A similar lawsuit was filed in Louisiana on Tuesday.
The pharmaceutical companies included in the lawsuit released the following statements:
Janssen Pharmaceuticals, Inc.: "We recognize opioid abuse and addiction are serious public health issues, and are committed to being part of the ongoing dialogue and to doing our part to find ways to address this crisis. Our actions in the marketing and promotion of these medicines were appropriate and responsible. The labels for our prescription opioid pain medicines provide information about their risks and benefits, and the allegations made against our company are baseless and unsubstantiated. In fact, our medications have some of the lowest rates of abuse among this class of medications."
Purdue Pharma: "We are deeply troubled by the prescription and illicit opioid abuse crisis, and we are dedicated to being part of the solution. As a company grounded in science, we must balance patient access to FDA-approved medicines, while working collaboratively to solve this public health challenge. Although our products account for less than 2 percent of the total opioid prescriptions, as a company, we've distributed the CDC Guideline for Prescribing Opioids for Chronic Pain, developed three of the first four FDA-approved opioid medications with abuse-deterrent properties and partner with law enforcement to ensure access to naloxone. We vigorously deny these allegations and look forward to the opportunity to present our defense."
Allergan: "It is important to put into perspective Allergan’s role regarding opioids. Allergan’s three legacy opioid products – Norco, Kadian and Fiorinal with Codeine — account for less than 0.04 percent of all opioid products prescribed in 2017 in the U.S. These products came to Allergan through legacy acquisitions and have not been promoted since 2013, in the case of Kadian and Fiorinal, and since 2003, in the case of Norco. Allergan has a history of supporting — and continues to support — the safe, responsible use of prescription medications. This includes opioid medications, which when prescribed and used responsibly, play an appropriate role in pain relief for millions of Americans."
Teva Pharmaceuticals: "Teva is committed to the appropriate use of opioid medicines, and we recognize the critical public health issues impacting communities across the U.S. as a result of illegal drug use as well as the misuse and abuse of opioids that are available legally by prescription. To that end, we take a multi-faceted approach to this complex issue; we work to educate communities and healthcare providers on appropriate medicine use and prescribing, we comply closely with all relevant federal and state regulations regarding these medicines, and, through our R&D pipeline, we are developing non-opioid treatments that have the potential to bring relief to patients in chronic pain. Teva also collaborates closely with other stakeholders, including providers and prescribers, regulators, public health officials and patient advocates, to understand how to prevent prescription drug abuse without sacrificing patients’ needed access to pain medicine."
Endo Pharmaceuticals and Endo Health Solutions declined to comment.
Amerisource, Cardinal Health and Johnson & Johnson did not respond for comment.
Kara Berg: 618-239-2626, @karaberg95
This story was originally published March 5, 2018 at 7:33 AM with the headline "Opioid-addicted baby at center of class-action lawsuit in Southern Illinois."